Glymphatic. I stumbled over the term glymphatic system last week and briefly had to pause. The concept is now widely used and feels intuitive, but it did not exist when I was in medical school. The term was only introduced in 2012 to describe a glia-dependent, perivascular clearance pathway in the brain. And yet, everything it refers to, including perivascular spaces, astrocyte endfeet, cerebrospinal fluid flow, interstitial clearance, was already in the textbooks. All the parts were familiar to me, but the label was new. That moment of confusion stayed with me: neuroscience moves forward not just through data, but also through names.
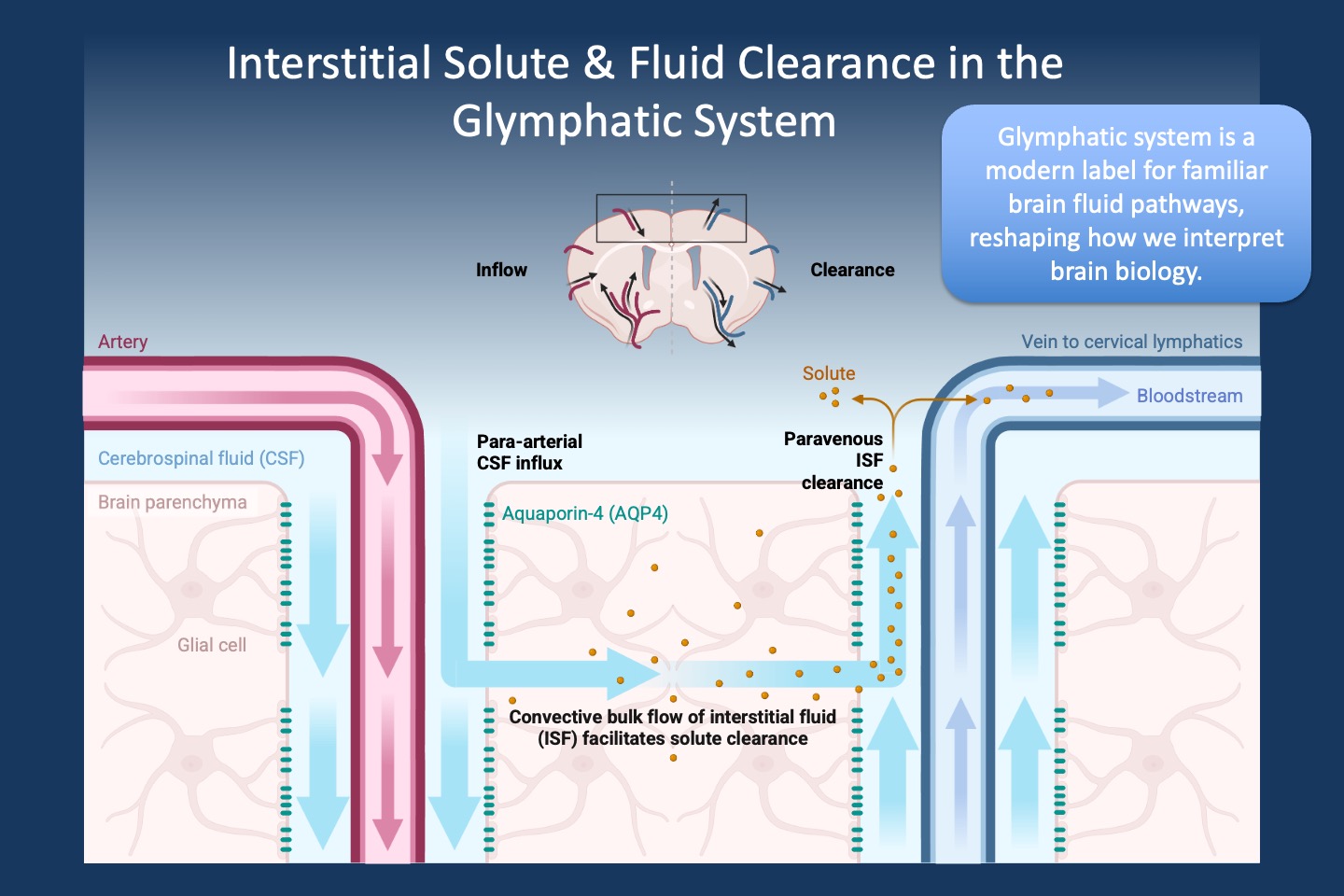
Figure 1. A modern map for an old pathway. Schematic of interstitial solute and fluid clearance in the brain as conceptualized in the glymphatic model. Cerebrospinal fluid (CSF) enters along para-arterial spaces, exchanges with interstitial fluid through astrocyte endfeet enriched in aquaporin-4 (AQP4), and exits along paravenous routes carrying metabolic waste toward the bloodstream and cervical lymphatics. While the anatomical components — perivascular spaces, astrocytes, CSF flow, and interstitial solute movement — have long been recognized, the “glymphatic system” framework, introduced in 2012, integrates these elements into a unified clearance pathway. This model has been influential in linking sleep, fluid dynamics, and neurodegeneration, even as the precise mechanisms and relative contributions of bulk flow versus diffusion remain under active investigation.
When a name creates a system. The concept of the glymphatic system was introduced as a glia-dependent, lymphatic-like clearance pathway in the brain. This new label did something powerful. It brought anatomy, physiology, sleep biology, and neurodegeneration into a single framework. Suddenly, CSF clearance was not just background plumbing but a potential key factor affecting health and disease. For example, neurodegeneration research reframed protein accumulation as a drainage problem. The term caught on.
But was it a discovery or a reframing. No new anatomical structure appeared when the word glymphatic entered the literature. Perivascular pathways had already been known for some time, including components such as CSF–interstitial exchange, astrocyte endfeet, and AQP4 polarization. But the glymphatic label suggested a coherent system with directional flow, closer to the lymphatic system than to uncoordinated perivascular diffusion. Whether that framing is biologically precise remains to be seen. What is clear, however, is that this new name reshaped the questions we ask.
Metaphors, models, and breakthroughs. Advances in science happen through all three, but they refer to different concepts. A breakthrough reorganizes reality and makes old explanations feel incomplete. A model explains a subset of observations and makes it possible to generate predictions. A metaphor helps us talk about complex biology in a way that is intuitive. Trouble begins when metaphors are mistaken for mechanisms. Neuroscience has been here before.
E–I imbalance in epilepsy. The epilepsy field has its own powerful metaphor: the idea that seizures arise from an imbalance between excitation and inhibition. This framework traces back to mid-20th century neurophysiology, when intracellular recordings first distinguished excitatory and inhibitory postsynaptic potentials and defined glutamate and GABA as the major excitatory and inhibitory neurotransmitters in the brain. The concept of E–I imbalance is grounded in physiology and remains highly informative. But as a general explanation for genetic epilepsies, it is insufficient. Many epilepsy genes do not simply push networks toward excitation or inhibition. They affect development, synaptic maturation, transcription, channel trafficking, and circuit formation. Some mutations impair inhibition in one region while reducing excitation in another. The E–I imbalance metaphor is useful, but it does not capture the full story of how networks become prone to generating seizures due to genetic changes.
Mirror neurons. Here is another example. Mirror neurons emerged in the early 1990s from single-cell recordings in macaque premotor cortex. Researchers observed neurons that fired both when a monkey performed an action and when it watched another perform the same action. This was a striking finding. From there, the concept expanded rapidly. Mirror neurons were invoked to explain empathy, language evolution, autism, and even the basis of human culture. These interpretations often outpaced the initial evidence, even as the primary research had already moved on.
Labels generate momentum. A good new label lowers barriers and connects fields. For example, it may give imaging researchers, molecular biologists, and clinicians a shared story. It also makes grant aims more legible and research programs more fundable. In that sense, even imperfect concepts can be productive, as they act as scaffolds. But eventually, the structure underneath has to stand on its own.
Conceptual narrowing. Once a metaphor becomes widely used and textbook-ready, it can quietly narrow thinking. If clearance is glymphatic, we may underappreciate vascular wall pathways or the role of diffusion. If epilepsy is E–I imbalance, we may struggle to integrate development, glia, or homeostatic plasticity. If social cognition is reduced to mirror neurons, we may miss network-level dynamics. The language we use does not just describe biology; it also shapes how we think about what is possible.
What this means for epilepsy genetics. The epilepsy genetics field has long organized many genetic epilepsies under the banner of channelopathies. This remains a powerful framework. Sodium, potassium, and calcium channel genes clearly link ion flux to neuronal excitability. The term helped unify disorders, guide functional studies, and frame therapeutic strategies. But even here, the label is a model rather than a complete description. Ion channels do not only set membrane potentials. They also influence development, synaptic release, dendritic integration, and network wiring.
Channelopathy as model, not destiny. Consider conditions due to disease-causing variants in CACNA1A. It is natural to classify them as calcium channel disorders. But CACNA1A also sits at the heart of neurotransmitter release and synaptic plasticity. The same condition could just as reasonably be conceptualized as a synaptic disorder, a circuit disorder, or a developmental disorder, depending on the lens we choose. Each label highlights one aspect of CACNA1A and hides others. Our narrative changes, but the underlying biology remains the same.
Stories we tell ourselves. Science runs on stories. We tell them to make sense of complexity and to connect data points. But the best stories are not permanent or set in stone. They are useful without being absolute. The glymphatic system, E–I imbalance, mirror neurons, and channelopathies are initial drafts that help us move forward. Over time, we may need to revise the narrative as the data demand it. The challenge is to use these conceptual tools to build connections and generate hypotheses while remembering that the underlying reality is always more complex than the story.
